For chemists needing to purify natural product extracts or synthesis reaction mixtures flash chromatography is typically the tool of choice. In previous posts I have discussed various ways to optimize the purification to obtain the highest purity compounds with maximum load in minimal time.
Sometimes, though, chemistry gets in the way in the form of solubility issues. When this happens most often dry loading is recommended for these sample types. In this post I will show the impact various dry load sorbent options have on chromatography.
Most chemists I know like to simplify their workflow, myself included. When it comes to chromatography that usually means a liquid injection of their crude material. However, liquid loading is not always an option due to dissolution solvent solubility/strength concerns.
In previous posts I have discussed the impact that the sample dissolution solvent choice can have on the purification quality. Choose a solvent that is too strong, chromatographically speaking, and you get poor separations and can get very broad peaks and even selectivity changes, not ideal. Usually, if the dissolution solvent is stronger than the “strong solvent” in your method you can have chromatographic issues, especially as injection volume increases.
So, how do we get good purification results if the only solvent that works is too strong for the purification method? Dry loading.
With dry loading you mix your dissolved sample with an adsorbent and evaporate the slurry to dryness. Once dry, you transfer the adsorbent into another vessel which is placed in-line with the main purification column, Figure 1. This is a proven formula for success but, like anything else, there are ways to improve dry loading.
-3.jpg?width=478&name=Dry-load%20(1)-3.jpg)
Figure 1. In-line dry sample loading (dry loading) often eliminates purification issues related to strong injection solvents.
One of the keys to successful dry loading is the sorbent. Most chemists will choose silica if normal-phase is to be used and C18 if reversed-phase is the mode, but there are other sorbent choices. For now, though, I want to focus on normal-phase.
Silica is a very good sorbent for dry loading, especially if you want to retain highly polar compounds also known as baseline material (i.e. compounds that do not move from the spotting origin in TLC). Using silica for dry loading can improve the actual separation performance because it protects the main silica flash cartridge from the very polar impurities.
There are other materials that are used which many chemists prefer. Among them are diatomaceous earth (e.g. ISOLUTE® HM-N), Florisil®, and alumina.
Think of diatomaceous earth more as a sponge than an adsorbent. Most compounds will only “bind” to it once the solvent has evaporated. Once rewet, the molecules will solubilize and pass into silica column. So, if you want to elute everything, this is the right sorbent.
Florisil is synthetic magnesium silicate and commercially available from many sources. Many chemists prefer it to silica and diatomaceous earth because of its high loading capacity and fast desorption kinetics.
Alumina is another effective dry loading media as it binds compounds well but not as strongly as silica and is more inert than silica from a reactivity perspective.
To highlight how these sorbents perform some colleagues of mine performed an evaluation of several of these sorbents for dry loading in a normal-phase purification of a 3-component mix (1-nitronaphthalene, 2-nitroaniline, and 3-nitroaniline, all dissolved in acetone). The results below show the potential improvements to flash purification achievable by dry loading with various sorbents. All the columns used were 25 gram Biotage® SNAP Ultra cartridges.
Figure 2 shows the chromatographic result when a chromatographically strong solvent at too large of a volume is used. The result is a separation that has distorted peak shape and will provide poor purity fractions.
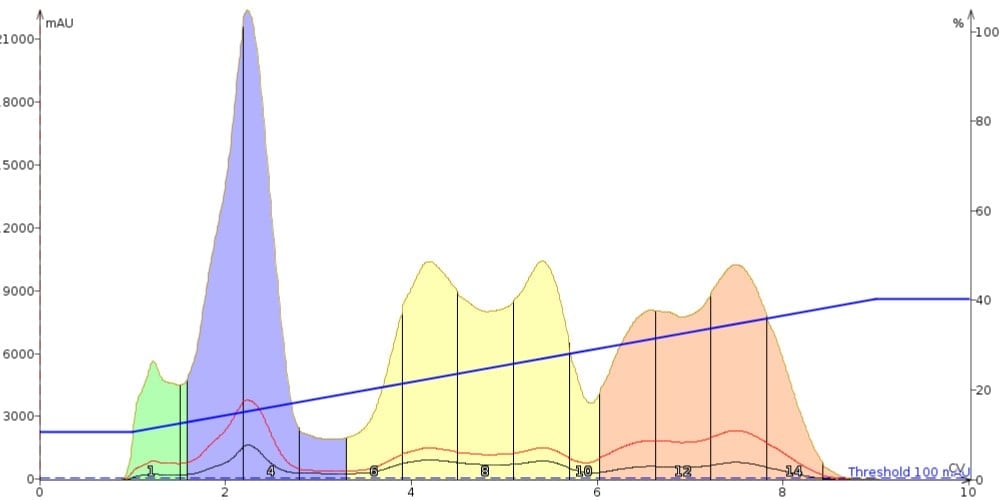
Figure 2. Impact of a dissolution solvent too strong for the purification is seen with peak de-banding (multiple peaks for the same compound).
In Figure 3 we see the impact on the separation from using silica as the dry load sorbent with the sample load. The result shows the overall separation and the amount of resolution between compounds 2 and 3 has improved dramatically compared to Figure 2.
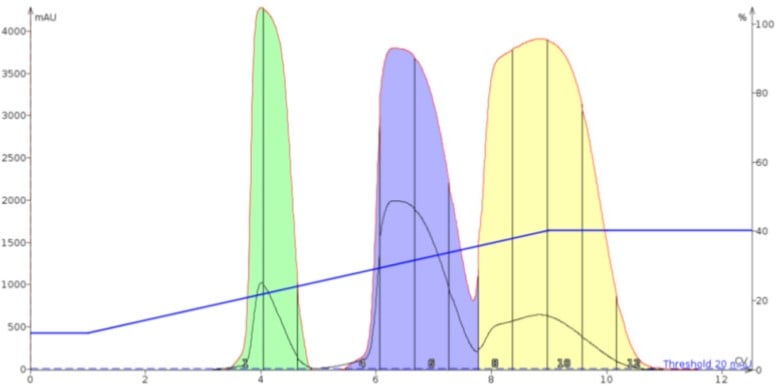
Figure 3. Dry loading with standard silica (50 µm) improves the separation compared to the liquid load.
Figure 4 shows us the results from using Florisil as the dry load sorbent. With Florisil we do see a better overall separation with sharper, better resolved peaks. The other take away from this example is that the sample components eluted earlier than when silica was used as the dry load sorbent. This is due to Florisil's lower adsorptive capacity which provides faster desorption kinetics.
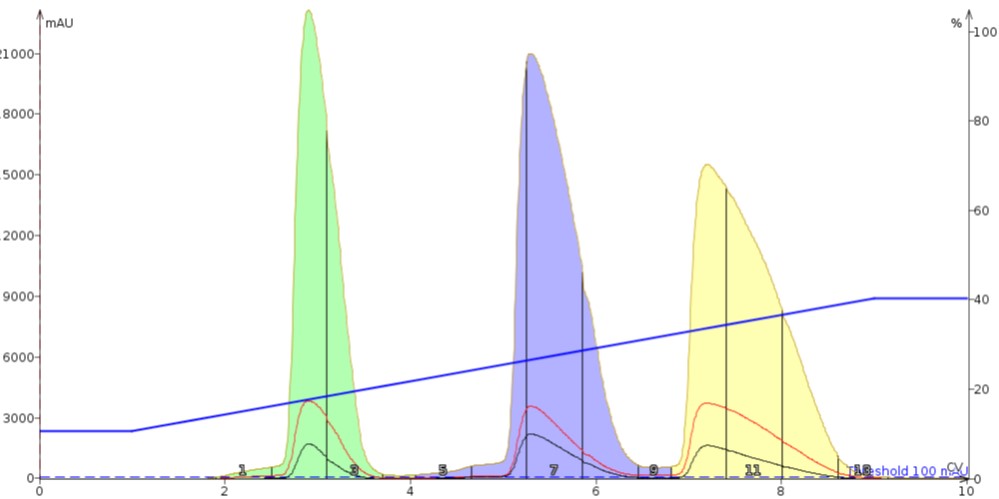
Alumina, as seen in Figure 5, also shows utility as a dry load sorbent improving the separation compared to both the liquid load and even silica. As with Florisil, alumina improved resolution and reduced elution times compared to silica.
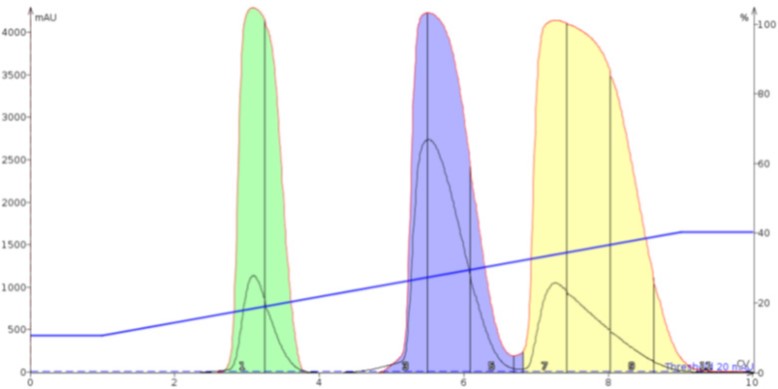 Figure 5. Alumina's inert, high capacity surface shows it has utility as a dry loading sorbent improving the separation compared to silica.
Figure 5. Alumina's inert, high capacity surface shows it has utility as a dry loading sorbent improving the separation compared to silica.The impact of using ISOLUTE® HM-N, a type of diatomaceous earth, is seen in Figure 6. The separation result is on par with alumina showing similar separation performance and runtime.
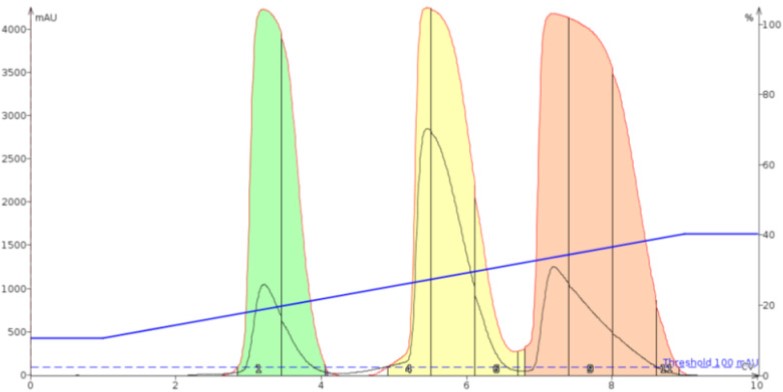
Figure 6. ISOLUTE® HM-N provides an excellent separation when used as a dry loading sorbent.
So, taking a look at the original issue - poor chromatography due to the use of an "incompatible" solvent the solution is dry loading. Your choice of dry load sorbent can impact the separation as well as seen in the results above. Though silica works, you may want to consider alternative media for dry loading such as Florisil, alumina, or ISOLUTE HM-N.
For more information on flash chromatography, please download our whitepaper - Successful Flash Chromatography.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership