Recently, I posted an article explaining why high performance TLC plates are not needed for method development for high-performance flash chromatography. Based on some excellent feedback, I see a need to discuss silica chemistry and its impact on chromatography.
In a nutshell, no two silica brands are the same. Sure, many have the same physical properties, such as surface area and porosity, but the surface chemistry between silica brands is almost always different. While the surface of silica particles is comprised primarily of hydroxylated silicon (Si-OH), not all of that chemistry is the same. According to Ewe Neue in his text “HPLC Columns – Theory, Technology, and Practice (Neue, 1997)”, there are several different silanol types including single silanols (Si-OH), silane diols or geminal silanols (HO-Si-OH), and bridged silanols or silyl ethers (Si-O-Si), Figure 1.
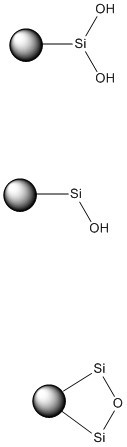
Figure 1. Structures of the three silanols types types. Top - geminal. Middle - single. Bottom - silyl ether.
Single and geminal silanols are quite polar and can be acidic. They are good at attracting polar compounds, organic bases, and other nucleophiles. Silyl ethers, on the other hand, are relatively hydrophobic and not very reactive.
So, how do the differences noted above impact what you do with flash chromatography? Well, consider the interactions taking place between your compound and the silica chemistry. Let's say you have a compound in your sample that is well retained on a silica TLC plate that happens to contain a high silyl ether percentage and you transfer the method to a flash column packed with a high single or geminal silanol content. With that scenario, it is very likely that your compound will elute later than you would expect from the flash cartridge. In fact, you may need a very different, more polar solvent system to perform the purification.
The ratio of these three silanols types impacts selectivity as well. Using unmatched TLC and flash silica has even caused compounds to change their elution pattern and/or selectivity.
I did not believe this early in my career but just one sample convinced me it is true. The sample was a mixture of organic dyes and the eluent was 100% toluene for both the TLC and the flash purification. Though I do not have a picture of the resulting flash chromatography, I do have the TLC data and the actual flash elution volumes to share, Table 1.
| Biotage | Competitor | Flash Elution | ||||
| TLC | TLC | volume | ||||
| Rf | CV | ΔCV | Rf | CV | ΔCV | CV |
| 0.84 | 1.19 | 0.82 | 1.22 | 1.25 | ||
| 0.34 | 2.94 | 1.75 | 0.33 | 3.03 | 1.81 | 3 |
| 0.18 | 5.56 | 2.61 | 0.25 | 4 | 0.97 | 5.5 |
| 0.1 | 10 | 4.44 | 0.09 | 11.11 | 7.11 | 9.25 |
What this data shows is that the more polar the compound (the lower the Rf), the more likely that a difference in retention and selectivity will be encountered when silica chemistry differs. And remember, low Rf values tend to provide better separations so small Rf differences can cause big column volume (CV) and ΔCV shifts.
In the example above, the ΔCV for the last two compounds varies significantly between the Biotage plate and another supplier's plate. Purifying the sample in a flash column packed with Biotage silica (Biotage® KP-Sil) provided elution column volumes with better correlation to the Biotage TLC plate than with the competition's plate.
Yes, I know you probably have a common TLC plate supplier and perhaps you have not experienced this problem so why change brands? Well that is certainly your choice, but for me, I would rather not chance a problem occurring because it is just a matter of time before you have issues which may lead to confusion during purification when transferring your TLC-based method to flash.
Have you ever experienced an issue like this? Share your experiences.
For more information on flash chromatography, download our whitepaper Inspiring Productivity with Modern Flash Chromatography.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership