Varying the concentrations of mobile phase solvents during flash purification chromatography enhances the ability of the technique to effectively isolate the desired compound from reaction by-products and unconsumed reagents. Choosing how these concentrations will be varied over time has a significant effect on the purity and recovery of desired compounds.
What is the best starting strong solvent %? What is the ending strong solvent %? Should the mobile phase concentrations vary gradually in a linear manner or should they vary step-wise or something else altogether? Most separations are performed once, occasionally a handful of times. Because of this, spending effort optimizing a gradient is just not very productive unless there are aids in choosing the gradient profile that provides an effective purification with minimum effort.
Software in flash chromatography instruments, makes it simple to create a gradient. Now, what should that gradient look like?
In this post I compare isocratic, step, and linear gradients and provide some sage advice on choosing among them.
Isocratic (uses a constant solvent ratio throughout the purification)In flash chromatography several elution methods can be used to purify compound mixtures…
Linear gradient (gradually increases the strong solvent percent over time)
Step gradient (uses a series of increasingly stronger isocratic steps over time)
In a previous post I discussed how to use normal-phase TLC data (Rf and % strong solvent) to create linear and step gradients. While isocratic elution can be effective it does suffer from elution band broadening that diminishes sensitivity/detectability and increases the collected fraction volume.
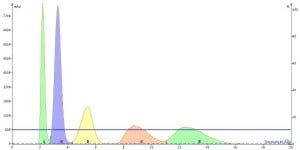 Figure 1. Isocratic elution of 100 mg of a 5-component mixture on a 10 g silica cartridge. Notice the increase in peak width with increasing retention.
Figure 1. Isocratic elution of 100 mg of a 5-component mixture on a 10 g silica cartridge. Notice the increase in peak width with increasing retention.
Linear gradients based on TLC data typically provide an improved separation as the gradient is defined by specific chromatographic parameters such as TLC strong solvent % and compound Rf values. If the compounds in the mix elute and separate with a specific % strong solvent on TLC, then a gradient based on the TLC plate’s data will be more accurate and elution more predictable than a generic 0-100% gradient. Some TLC to linear gradient capable flash systems (e.g. Biotage® Isolera) will even estimate sample loading!Linear gradients, on the other hand, minimize band broadening since the elution solvent strength is increasing during the purification. Linear gradients are simple and typically the default elution method for many flash systems. Isco, for instance, uses a default 0-100% linear gradient over 16+ column volumes (CV) for their purification methods; Biotage does the same for its cartridge-based default methods but over a shorter 13 CV. Though simple, this generic approach to chromatography is not always successful as the gradient slope may be too steep or too shallow to generate a suitable separation.
But even with data-derived linear gradients the separation of any pair of compounds may not be as good as you would like. This is due to the fact that the linear gradient is just that – linear. Linear gradients employ the same % strong solvent rate of change over the course of the separation which can force compounds to elute more quickly than desired generating unresolved peaks and blended fractions.
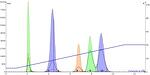 Figure 2. Linear gradient separation of 100 mg of a 5-component mixture using a 10 g silica cartridge. The linear gradient reduces the peak width, increased peak height, and has changed the amount of separation between each of the peaks.
Figure 2. Linear gradient separation of 100 mg of a 5-component mixture using a 10 g silica cartridge. The linear gradient reduces the peak width, increased peak height, and has changed the amount of separation between each of the peaks.
Perhaps a better alternative is a step gradient. As discussed in a previous post, a step gradient derived using solvent and Rf data from as few as two (2) TLC plates can be achieved. Step gradients have the potential to separate and elute each compound individually in minimal volume with maximum resolution which, in my experience, relates to increased loading capacity – another step gradient benefit.Employing an isocratic hold or pause can help but where in the gradient do you add it, and for what volume? Certainly, adding the hold during a peak’s detection won’t work as it is exiting the cartridge and entering the detector - it already eluting. The hold needs to be built into the gradient at least one (1) CV earlier. But this is all guesswork, not conducive to optimal time management and not guaranteed to work without multiple purification trial runs (who has the time?).
The chromatography examples in this post show a 5-component mix being purified using a 10 g silica cartridge first by an isocratic method (figure 1), then by a linear gradient (figure 2), and finally by a step gradient (figure 3). The biggest difference between them is the amount of purified material - 100 mg on both the isocratic and linear gradients but 200 mg with the step gradient, a testament to the power of the step gradient.
-1.jpg?width=724&height=362&name=5-component-step-gradient%20(1)-1.jpg)
Figure 3. Example of a TLC-derived step gradient separating 200 mg of the same 5-component mixture using a 10 g silica cartridge.
For my work I often will use TLC-based linear gradients for simplicity but if I want to maximize my compound purity or load capacity, I will develop a two or three TLC plates in parallel and create a step gradient.
If you're wanting to learn more about being successful in flash, read my white paper on the subject below!

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership