For most chemists, flash purification is a means to an end. In other words, it is a tool needed to purify and isolate one compound from a mixture of compounds so that the next reaction can occur with reduced by-product formation. Other than choosing between normal- or reversed-phase, there typically is not much thought put into cartridge selection, especially not related to stationary phase media porosity.
For most small molecules, this approach makes sense, but for larger molecules and very lipophilic compounds, factoring for media porosity should be included. In this post, I will discuss the impact media porosity can have on chromatographic performance.
Media porosity, why should I care? Well, most of the time you will not need to, since most small molecules make their way into the media’s pores (that is where the vast majority of the surface area lies). For bio-molecules such as peptides or polymers, which can vary in size (and shape), media porosity is an important factor to consider when developing a purification method - the larger the molecular weight (MW) or hydrodynamic volume, the larger the media porosity.
Normal-phase silica has, on average, 50 to 60 Å pores (and a surface area of ~500 m²/g). However, for small-molecule reversed-phase chromatography to be useful, the average pore size must be larger (90 to 120 Å). The reason for this is that the bonded phase (typically C18) takes up space on the silica surface and can block access to the pores (and the majority of the available surface area). So, using slightly wider pore diameter silica as a support for the bonded phase improves surface accessibility for most small molecules.
There are instances where a wider pore reversed-phase media is beneficial – highly hydrophobic compounds that are not well retained or separated using normal-phase chromatography.
Why is this? Because media pore size and surface area are inversely related – larger pores equals smaller surface area. Lower surface area equates to lower bonded phase content and, therefore, faster compound elution requiring less solvent, albeit, at lower loading capacity.
As an example, I purified a mixture of α-tocopherol and cholesterol using a conventional reversed-phase column (Biotage® Sfär C18, 12 gram, 18% carbon, 100 Å pores) and a 300 Å Biotage® Sfär Bio C18, 10 gram column (8% carbon). These compounds are very lipophilic and require copious amounts of organic solvent to elute from a conventional C18 column, Figure 1.
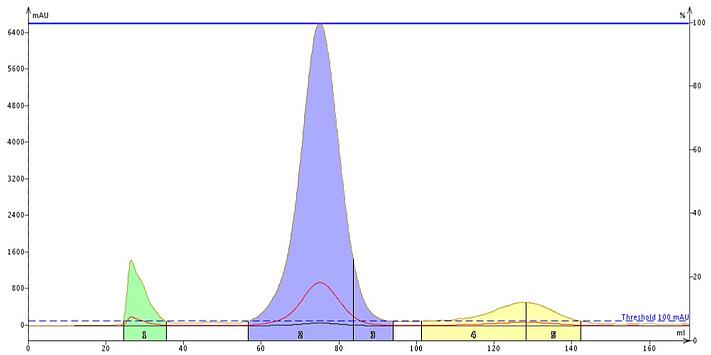 Figure 1. Standard C18 columns can require a lot of organic solvent to separate and elute hydrophobic sample components, which is not efficient. In this case tocopherol (blue) required 95 mL of pure methanol to elute off a 12 g c18 column while cholesterol required over 140 mL. Both tocopherol and cholesterol had peak volumes of ~ 40 mL (more than 2 column volumes).
Figure 1. Standard C18 columns can require a lot of organic solvent to separate and elute hydrophobic sample components, which is not efficient. In this case tocopherol (blue) required 95 mL of pure methanol to elute off a 12 g c18 column while cholesterol required over 140 mL. Both tocopherol and cholesterol had peak volumes of ~ 40 mL (more than 2 column volumes).
In comparison, using a lower carbon content, wider pore C18 reduces elution volumes and collection volumes in half with minimal separation loss at the same sample load, Figure 2.
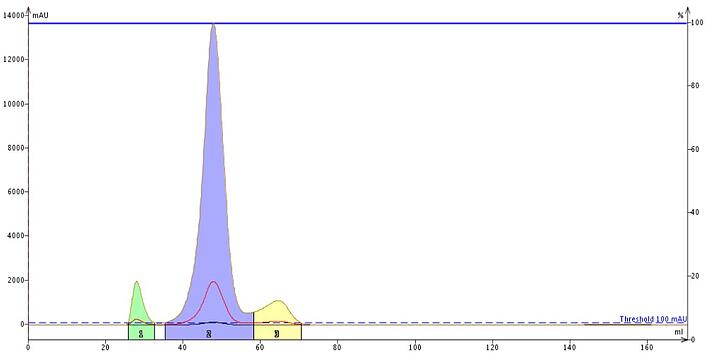 Figure 2. Purification of same sample using the wide pore Sfär C18 column with a lower % carbon reduces compound retention and solvent consumption. So, if you struggle with "sticky" compound purification, give wide-pore media a chance. You may be pleasantly surprised with the results.
Figure 2. Purification of same sample using the wide pore Sfär C18 column with a lower % carbon reduces compound retention and solvent consumption. So, if you struggle with "sticky" compound purification, give wide-pore media a chance. You may be pleasantly surprised with the results.
For more information on flash chromatography, please download our whitepaper - Enhancing Productivity with Modern Flash Chromatography.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership