For most organic reaction mixture purifications the process is fairly straightforward. Use hexane/ethyl acetate or, for polar compounds, DCM/MeOH. But what do you do if this doesn't work and your compounds are basic organic amines?
In this post, I re-examine the options available to achieve an acceptable organic amine purification when typical separation methods are insufficient.
In chromatography, the most important parameter to optimize is selectivity. Selectivity is the amount of separation between any pair of sample compounds. Therefore, it is beneficial to maximize selectivity (or separation) to ensure the best fraction purity and yield. Without selectivity, there is no separation.
Normally, the differences in sample compound chemistry along with the column media chemistry (stationary phase) and elution solvent chemistry (mobile phase) is enough to create selectivity and therefore a separation. However, sometimes the chemistry differences between individual sample compounds is slight or the compounds’ functionality is reactive or ionizable.
These situations complicate the separation and when they arise, method changes are needed. These changes can be in the form of the solvent system (changing solvents), the stationary phase (changing column media), or both.
Organic amines, especially heterocyclic, tertiary, and secondary amines that are basic can be quite challenging to separate and purify. If using typical normal-phase chromatography with silica columns the biggest problem is an acid-base interaction between silica (a BrØnsted acid) and the basic amines. This interaction can cause compound degradation, yield loss, and/or increased bandspreading. To counteract these problems you can employ one of the three options listed below.
A. Add a competing amine to the mobile phase
By incorporating a base in the elution solvent for both equilibration and the actual purification the acidic silica silanols are “neutralized” by the small, competing amine mobile phase modifier. Competing amines include ammonia, triethylamine, pyridine, ammonium hydroxide, etc. in a mobile phase of DCM/MeOH. This, however, can be challenging to get to work due to the strong displacement effects of both methanol and the added base, so I prefer option B.
B. Change column media
To stay in a normal-phase purification mode you can replace your silica column with one packed with either basic alumina or an amine-functionalized silica.
Amino-silica has the advantage of maintaining the same particle size as standard silica (alumina typically has a much larger particle size) but because it is bonded with an organic amine, the surface is now “basic” and organic amines separate using “softer and safer” solvents such as hexane/ethyl acetate or ethyl acetate/IPA – no added base is required.
In Figure 1, we show an example of a DCM/MeOH purification of three tricyclic amines. The elution gradient originally programmed (0-20% MeOH in DCM) was based on TLC data which showed a separation of the three compounds without any amine modifiers. So, using a silica column and no any modifier I used the suggested gradient. Well, the results are not what was expected or needed as nothing eluted using the programmed method. So, to try to elute the compounds I increase the methanol % which eventually eluted the compounds in a single peak.
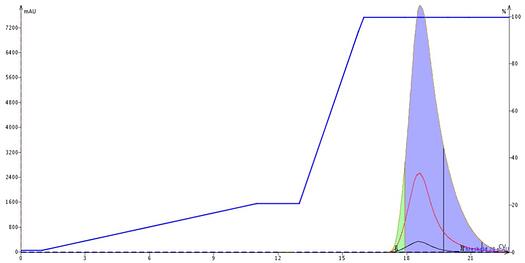
Figure 1. DCM-MeOH gradient of three basic tricyclic organic amines based on TLC data at 5% MeOH/95% DCM provides no separation of the compounds.
That result prompted me to switch from silica to an amine-functionalized silica. As can be seen below, this column chemistry works by providing a chromatographic environment much more conducive the organic amine purification and I was able to use hexane with ethyl acetate as my solvents, Figure 2.
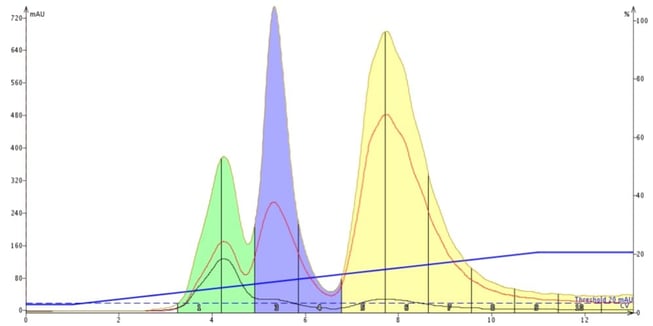
Figure 2. An amine-functionalized silica column (Biotage® KP-NH) successfully separates the three basic, tricyclic organic amines using a hexane/ethyl acetate gradient.
C. Use reversed-phase
Reversed-phase HPLC or UPLC is typically used to monitor a reaction’s progress. That being said, if it works on an analytical scale why not use it for preparative flash chromatography? It can be useful for polar, ionizable, and lipophilic compound purification.
Basic amine compounds are best retained and separated when the mobile phase pH is alkaline and in the free-base form. At high pH, these compounds become more lipophilic and therefore more retentive increasing the probability of a successful purification. To achieve this goal I like to employ the 2 pH rule which states to deprotonate an amine, adjust the solution pH to two units above the amine's pKa. For this I like to use a volatile base (easier to evaporate) at the 0.1% level in both the weak and strong solvents. Examples of volatile bases include...
i. Ammonium hydroxide
ii. Diethylamine (DEA)
iii. Triethylamine (TEA)
In Figure 3, we show an example where three organic amines, 4-dimethylamino antipyrine (pKa 9.6), phenylbenzyl amine (neutral), and amitriptyline (pKa 9.4) are separated using a gradient incorporating TEA as the base. By increasing mobile phase pH compound hydrophobicity is improved and retention increases compare to a neutral pH mobile phase or an acidic mobile phase. Another benefit to purifying amines in an alkaline solvent is you use more organic solvent (because of the increased compound hydrophobicity) which then is easier to evaporate post purification.
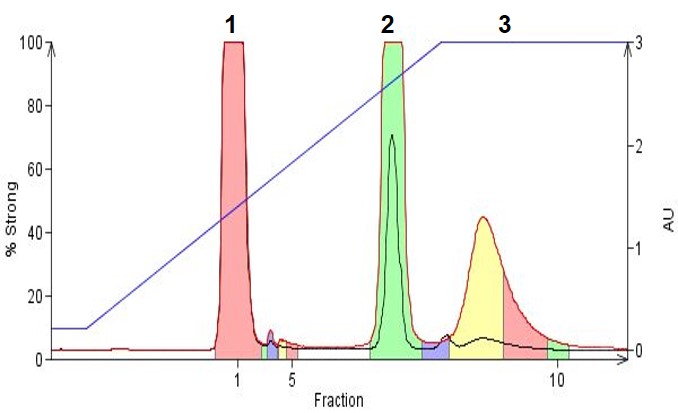
Figure 3. Organic amines purified using reversed-phase gradient flash chromatography. Solvents used were acetonitrile, water and 0.1% TEA. Peak 1 dimethylamino antipyrene, Peak 2 phenylbenzyl amine, Peak 3 amitriptyline.
So, the moral of the story is to create purification methods best suited for organic amines, choose an appropriate stationary phase (e.g. amino-functionalized silica, reversed-phase) and the right solvent conditions (pH, buffers, solvent chemistry/functionality) and you will improve your purification success rate.
Interested in learning more about purifying amines? Click the link below.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership