Acetone, as you know, is a terrific solvent. It dissolves many organic molecules, evaporates easily, is both water and organic soluble, and is cheap (relatively). These attributes tell me it should be a good polar modifier for normal-phase flash chromatography.
There is one attribute that keeps chemists from using acetone in chromatography - its strong UV absorbance above 250 nm which can mask compound detection and make UV-triggered peak fractionation a challenge, especially aromatic compounds. However, with some state-of-the-art flash systems, mobile phase UV absorption is zeroed in real time enabling the use of UV absorbing solvents such as acetone.
In this post I discuss some reasons to explore switching to acetone and how modern flash chromatography equipment deals the UV-absorbing mobile phase solvents.
Historically, ethyl acetate (EtOAc) has been the “polar” solvent of choice within normal-phase flash chromatography. Why? Because it is a good solvent for many organic compounds, evaporates easily, and has low UV absorbance above 250 nm where many organic compounds have their strongest UV absorption.
So, what are the downsides of using EtOAc? Well, I can think of only two concerns with EtOAc…
- -Price (~$40/Liter) in a 20-L drum
- -Chromatographic performance
I guess you would like some explanation for concern 2. Well, in my experience EtOAc works well but I find acetone works even better providing sharper peaks and improved resolution between them.
Acetone and EtOAc are in the same solvent selectivity class which means they are essentially interchangeable from a chromatography perspective. If you have a method that uses EtOAc, chances are you can replace it with acetone. Of course if your sample contains organic amines that can react with ketones, I would not make the switch.
Figure 1 below is a representative gradient separation using hexane and EtOAc. The chromatogram shows a good separation but not a complete separation, especially for the last two compounds.
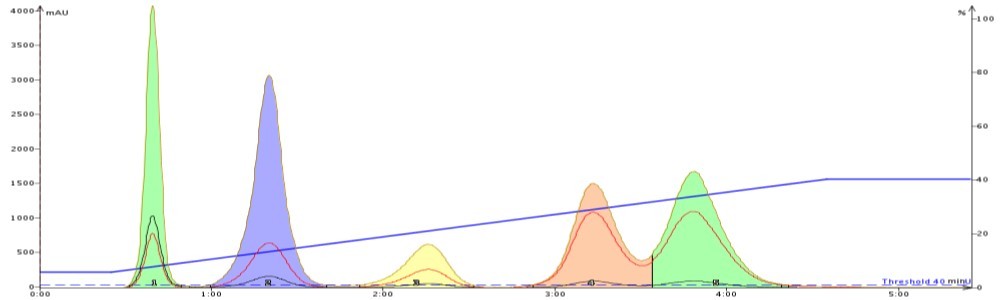 Figure 1. Hexane/ethyl acetate gradient separation of a 50 mg mix containing naphthalene, 1-nitronaphthalene, 3,5-dibenzyloxyacetophenone, butyl paraben and methyl paraben. A Biotage ZIP® 10 g cartridge operated at 36 mL/min was used. The last two compounds were not fully separated.
Figure 1. Hexane/ethyl acetate gradient separation of a 50 mg mix containing naphthalene, 1-nitronaphthalene, 3,5-dibenzyloxyacetophenone, butyl paraben and methyl paraben. A Biotage ZIP® 10 g cartridge operated at 36 mL/min was used. The last two compounds were not fully separated.
By just replacing EtOAc with acetone and running the same gradient, each peak sharpens which in turn improves the separation, Figure 2. The gradient does not show any UV absorption from acetone because the flash system I used, an Isolera™ Spektra from Biotage, incorporates a solvent UV absorption correcting algorithm which subtracts any solvent UV absorption during the gradient in real time.
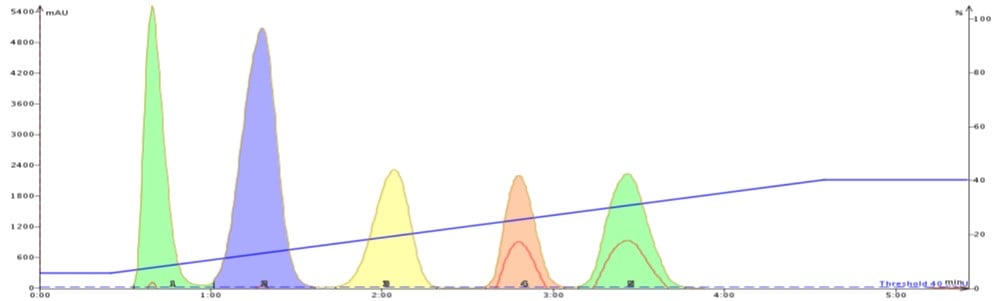
Figure 2. Hexane/acetone purification of the same 5-component mix using the same gradient conditions, cartridge, and 50 mg load shows a much improved separation, especially for the last two compounds. It is believed that acetone positively impacts the mass transfer of the more polar compounds slightly decreasing retention while reducing band-broadening. Reduced band broadening improves detection and minimizes fraction volume.
Now that the technical issue with detection is removed, the other motivation for changing to acetone is cost. I recently checked solvent pricing on-line and found HPLC grade EtOAc in a 20-L drum for $806 or ~$40/L. Acetone, I discovered, is priced 34% lower ($534) for the same HPLC grade, also in a 20-L drum (both solvents from Sigma-Aldrich).
Interesting. Acetone is less expensive and provides improved chromatography. Nice.
If you are interested in learning more about flash chromatography, please download our whitepaper Inspiring Productivity with Modern Flash Chromatography.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership